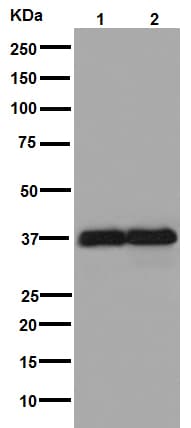
Loading control western blot

Sundaram P (2018) Protein stains and applications. Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Ni D, Xu P, Gallagher S (2017) Immunoblotting and Immunodetection. Key wordsīass JJ, Wilkinson DJ, Rankin D et al (2017) An overview of technical considerations for Western blotting applications to physiological research. Here, we present a rapid Western blot protocol, which combines fast blotting using the iBlot system and fast immunostaining utilizing ReadyTector ® all-in-one solution with the Smart Protein Layers (SPL) approach. To overcome these limitations, stain-free methods were developed allowing the combination of fluorescent standards and a stain-free fluorescence-based visualization of total protein in gels and after transfer to the membrane. Chemiluminescence-based methods are straightforward, but the detected signal does not linearly correlate to protein abundance (from protein amounts >5μg) and have a relatively narrow dynamic range. Conventional colorimetric staining tends to suffer from low sensitivity, limited dynamic range, and low reproducibility. For Western blotting several detection methods are available, e.g., colorimetric, chemiluminescent, radioactive, fluorescent detection. This approach is independent of a single loading control, and precision of quantification and reliability is increased. Another strategy uses total protein normalization where the abundance of the target protein is related to the total protein amount in each lane.
However, several studies have already shown that this is not always the case making this approach suboptimal. Usually, normalization of the target protein signal is performed based on housekeeping proteins (e.g., glyceraldehyde 3-phosphate dehydrogenase, ß-actin) with the assumption that those proteins are expressed constitutively at the same level across experiments. Major challenges for the reliable protein quantification by Western blotting are adequate data normalization and stable protein detection. Nevertheless, with the development of fast blotting systems and further development of immunostaining methods, a reduction of the processing time can be achieved. However, the whole procedure is often very time-consuming. It enables detection of a target protein based on the use of specific antibodies. XP is a registered trademark of Cell Signaling Technology, Inc.For the quantification of certain proteins of interest within a complex sample, Western blot analysis is the most widely used method.

Want more information about running a successful western blot? For most target proteins and model systems this is optimal when probing with CST antibodies. We recommend loading 20–50 μg of total protein per lane on a mini gel. Or worse yet, a disgruntled third reviewer may require new experiments to justify publication. As the old carpenter says, “measure twice, cut once.” Some care in advance can prevent having to go back and repeat those blots to get truly quantitative data just before publication.
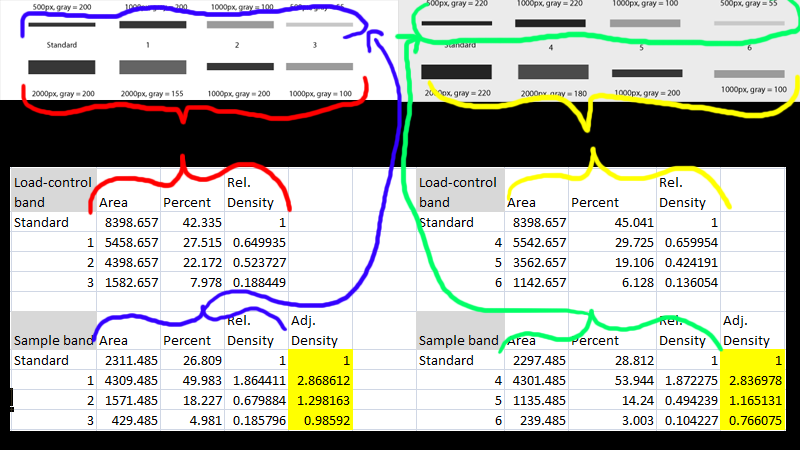
So, unfortunately, it may be necessary to run some dilutions of your precious samples to determine the dynamic range of the protein of interest and the loading control. In general, the evaluation of several loading controls is useful to identify at least one, and better yet several, that demonstrates linearity within the range of your protein of interest. As a result, many controls may have to be considered since no single protein will meet all these criteria for every conceivable experiment. Often, some preliminary scouting is required to obtain appropriate protein concentrations. It is the final criterion that is most frequently violated, with scientists making futile attempts to measure the shape and density of the band rather than running yet another blot. Third, and most importantly, the signal detected for both the loading control and the protein of interest must be in a linear range, otherwise you’ll find yourself with burnt-out bands that you’ll be unable to quantify. Second, the molecular weight of the control must be different from the protein of interest. First, the protein level of the loading control must not be altered by experimental conditions. For those who wish to bring a measure of rationality to the western blot data in the literature, loading controls can induce a fair amount of hand wringing.
